About
 Cardiolite® is a technetium-labeled single photon emission tomography (SPECT) myocardial perfusion imaging (MPI) agent used for identifying and managing patients with known or suspected coronary artery disease (CAD) and has been used to image tens of millions of patients since 1991.1,2
Cardiolite® is a technetium-labeled single photon emission tomography (SPECT) myocardial perfusion imaging (MPI) agent used for identifying and managing patients with known or suspected coronary artery disease (CAD) and has been used to image tens of millions of patients since 1991.1,2
Cardiolite® was the first technetium-labeled myocardial perfusion imaging tracer to provide physicians with prognostic information helpful in making patient management decisions related to CAD.1
Questions? For more information, please contact us.
IMPORTANT SAFETY INFORMATION
INDICATIONS AND USAGE
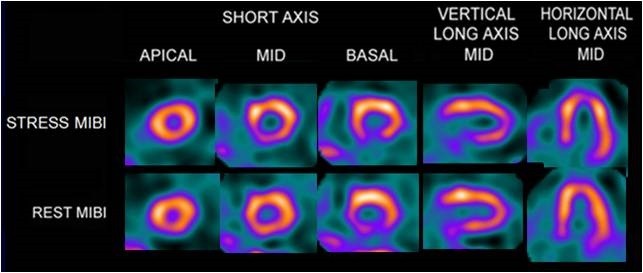
Myocardial Imaging: Cardiolite® (Kit for the Preparation of Technetium Tc99m Sestamibi for Injection), is a myocardial perfusion agent that is indicated for detecting coronary artery disease by localizing myocardial ischemia (reversible defects) and infarction (non-reversible defects), in evaluating myocardial function and developing information for use in patient management decisions. Cardiolite® evaluation of myocardial ischemia can be accomplished with rest and cardiovascular stress techniques (e.g. exercise or pharmacologic stress in accordance with the pharmacologic stress agent’s labeling).
CONTRAINDICATIONS
None known.
IMPORTANT SAFETY INFORMATION
Cardiolite® has been rarely associated with acute severe allergic and anaphylactic events of angioedema and generalized urticaria. In some patients the allergic symptoms developed on the second injection during Cardiolite® imaging. The most frequently reported adverse events include headache, chest pain/angina, ST segment changes on ECG, nausea, and abnormal taste and smell.
Infrequently, death has occurred 4 to 24 hours after Tc99m Sestamibi use and is usually associated with exercise stress testing (See Section 5.2). Pharmacologic induction of cardiovascular stress may be associated with serious adverse events such as myocardial infarction, arrhythmia, hypotension, bronchoconstriction and cerebrovascular events.
WARNINGS AND PRECAUTIONS
In studying patients in whom cardiac disease is known or suspected, care should be taken to assure continuous monitoring and treatment in accordance with safe, accepted clinical procedure.
Caution should be exercised and emergency equipment should be available when administering Cardiolite®.
Before administering Cardiolite® patients should be asked about the possibility of allergic reactions to either Cardiolite® or Miraluma®. Miraluma® is an identical compound used in breast imaging.
The contents of the vial are intended only for use in the preparation of Technetium Tc99m Sestamibi and are not to be administered directly to the patient without first undergoing the preparative procedure.
References: 1. Cardiolite® [package insert]. N. Billerica, MA: Lantheus Medical Imaging. 2. Data on file. Lantheus Medical Imaging, Inc.
 |
This site is intended for U.S. Healthcare Professionals only. PM-US-CA-0018-V2 October 2024 |
